
Electron Affinity:Įlectron Affinity is a property describing the tendency of a neutral atom to gain an electron, in order to form a negative ion. Note: This applies to groups 1,2 (metals) and to groups 15,16, and 17 (non-metals). The most highly reactive element to be observed is cesium, as it spontaneously reacts with air and water! On the other hand, reactivity in non-metals increase further right down a period, and further up a group. The reactivity of metals increases further left along a period, and further down a group. Reactivity is dependent on the classification of an element (metals and non-metals), as they both have differing periodic trends. These are factors that affect the interactions of electrons that chemical reactions undergo. This property is dependent on characteristics such as electronegativity and ionization energy. Reactivity describes the ability of a molecule or atom to undergo a chemical reaction, followed by a release in energy. This allows for electrons to be retained as a result of these attractive forces. 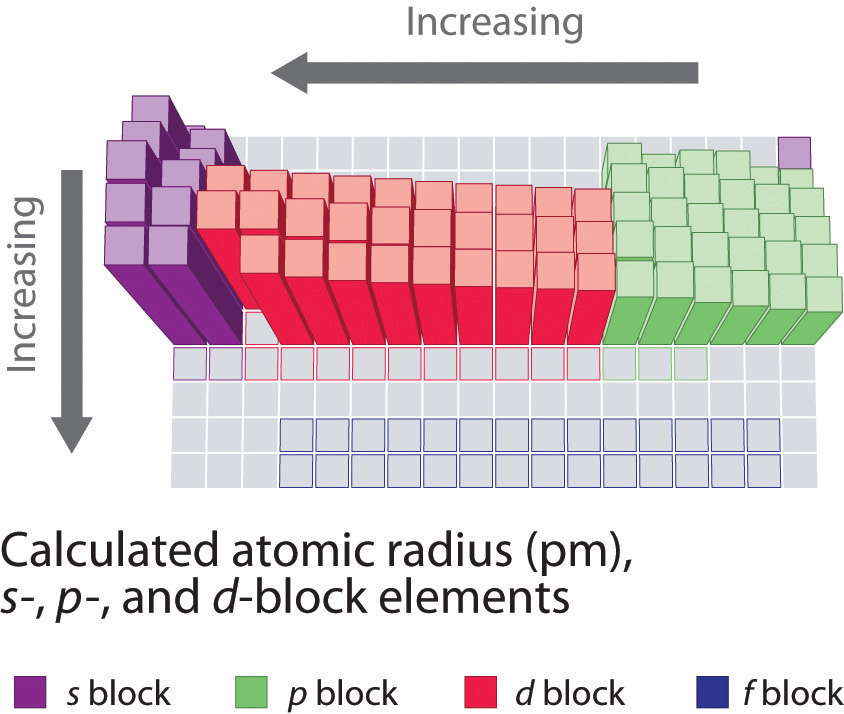
An example of high ionization energy is in helium, as it has a very small atomic radius with +2 charge present in the nucleus. The Ionization energy increases further right down the period, and higher up a group as more energy is required to remove a tightly bounded electron from the atom. This property is describing the difficulty of removing an electron. The minimum amount of energy required to remove an electron from a neutral atom’s outermost electron shell in the gaseous phase is called the ionization energy. Note: This periodic trend excludes the Noble gases (Group 18). Note that the noble gases do not have an electronegativity. 
This is observed through Periodic Trends, as the further right of a period, and higher up a group are observed to have increased values in electronegativity. This scale ranges from 0.7 to 4.0, where cesium is the least electronegative element, and with fluorine being the most electronegative element. It was created by measuring the bond energy of the different elements joined by covalent bonding. The scale that was formed in order to measure this property is the Pauling scale. Which properties can be identified using periodic trends? Electronegativity:Įlectronegativity is a property that measures the tendency of an atom to attract electrons to form a bond. We discuss trends for properties such as electronegativity, atomic radius, first ionization energy, reactivity and electron affinity. This is due to the structural similarities’ elements have within a period or family that allows for these trends to take place. These trends have allowed scientists in the past to predict certain characteristics of unknown elements. Why is it so much larger? Because the first two electrons are removed from the 3 s subshell, but the third electron has to be removed from the n = 2 shell (specifically, the 2 p subshell, which is lower in energy than the n = 3 shell).Periodic trends are observable patterns in the properties of an element that are dependent on its position in the Periodic Table. The third IE, however, is over five times the previous one. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
